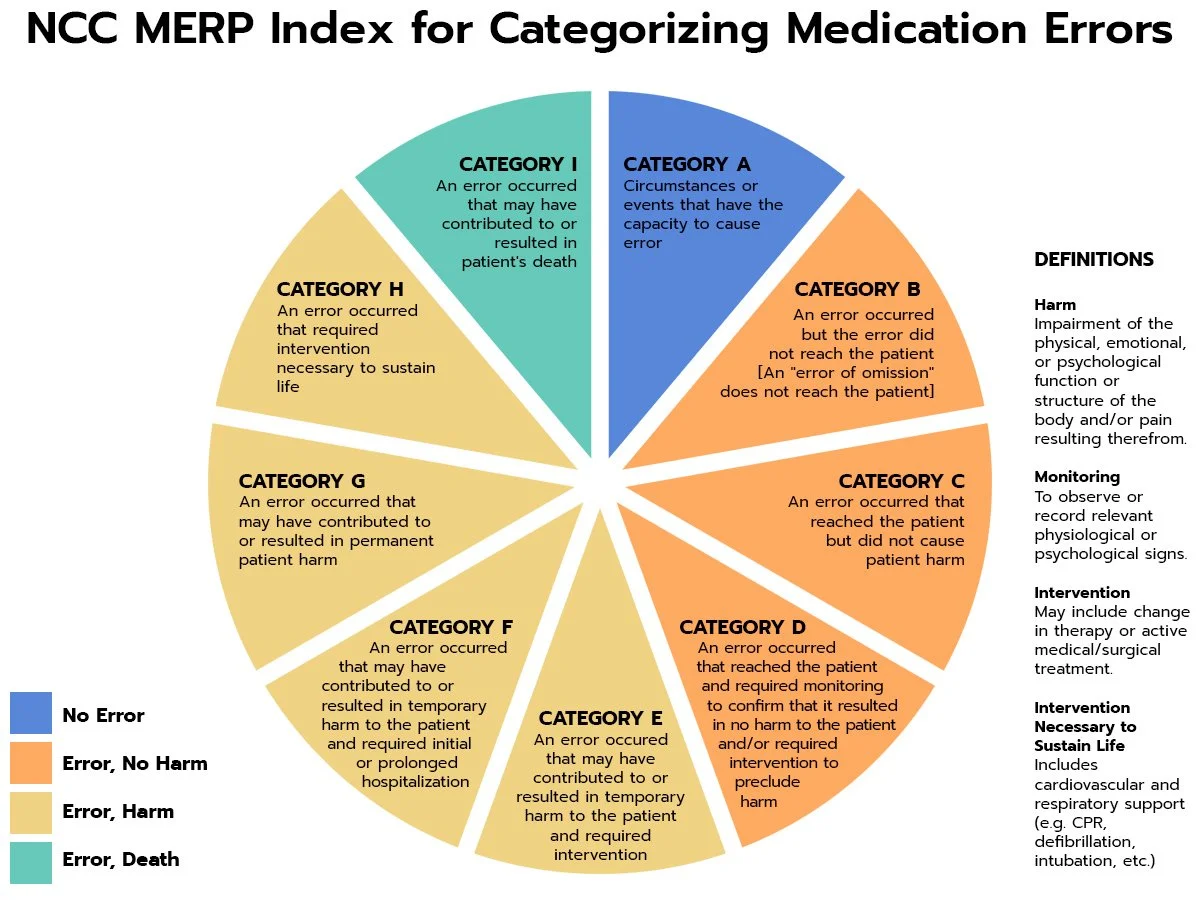
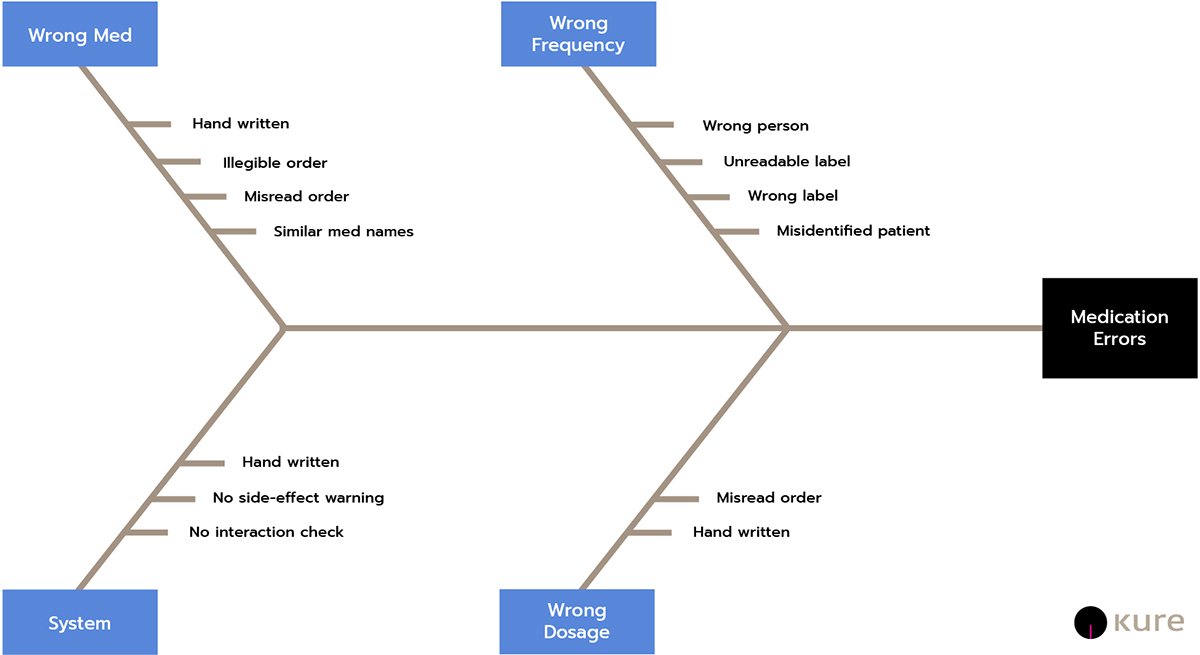
Reducing Prescription and Medication Issues With Process Optimization
Instant Insight:
Prescribing errors represent a billion dollar liability for healthcare organizations
Human error, including physician distraction, is a substantial cause of errors
Prescribing errors may be traced to systemic failures
The “Blame Game” of error responsibility must be replaced with a team approach
A thorough, system-wide process review can result in significant improvement
The Impact of Prescribing Errors
Physicians, medical staff and hospital organizations have come under increased scrutiny and regulation regarding prescribing practices and patient medication issues. Physician licensing, insurance coverage, and facility/pharmacy liability for errors are all part of the prescribing practice quotient.
In California, for example, since 2015 the Medical Board has expended substantial money and resources on investigating physician prescribing through the state’s CURES data system¹, initiating accusations against physicians based on prescribing patterns deemed inappropriate by Board-employed experts without a patient or institutional complaint.
Enacted in 1997, the CURES system was originally intended to simply monitor and provide prescribing information and determine the nature and amount of prescription drug use. In 2015, however, then-Attorney General Kamala Harris saw prescription drug abuse as a potent political issue and pushed for legislation to enact CURES-2, a much broader data collection system in order to “lead the fight against prescription drug abuse.”²
“. . .the total cost of looking after patients with medication-associated errors exceeds $40 billion each year. In addition to the monetary cost, patients experience psychological and physical pain and suffering as a result of medication errors. Finally, a major consequence of medication errors is that it leads to decreased patients’ satisfaction and a growing lack of trust in the healthcare system.⁴”
Harris initially assured physicians and pharmacists that the new version of CURES would simply enable them to better track prescriptions and help identify patient drug abusers. However, within a few short years CURES was weaponized by the Medical and Pharmacy Boards as an enforcement tool to be used against providers rather than the drug-abusing patients. As a result, many healthcare professionals became leery of the expanded CURES reporting requirements, with the large number of CURES’ generated accusations, and therefore less inclined to be cooperative.
Along with the expanded CURES program, California’s Medical Board also pursued a controversial Death Certificate Program in which coroners were required to report all patient deaths to the Medical Board for review to determine if improper or inappropriate prescribing had occurred and/or contributed to the death.³ Hundreds of physicians were targeted and subjected to investigation and possible accusations, but only a minority of cases actually involved drug overdoses. (The program has since been narrowed in scope to opioid overdose cases.)
However, because California is considered by many to be a lodestar in healthcare regulation, healthcare professionals in other states will undoubtedly face increased prescribing regulations, restrictions and government scrutiny in the coming months and years.
Prescribing errors represent a billion dollar liability issue for healthcare systems nationwide. These errors can result in significant morbidity and mortality. While the problem has been recognized worldwide, the best way to address these concerns remains the development of a multifaceted strategy for education and prevention. Teamwork, not individual action or blame, must become the institutional focus if prescribing errors and costs are to be reduced. A culture of safety and clear communication must be encouraged through systemic review and change.⁵
A National Center for Biotechnology Information (NCBI) article recently described common dispensing errors and prevention.⁶ Among the plethora of prescribing issues facing healthcare in 2021, NCBI lists the following as primary causes for concern:
“Prescribing errors represent a billion dollar liability issue for healthcare systems nationwide. Teamwork, not individual action or blame, must become the institutional focus if prescribing errors and costs are to be reduced.”
Medication errors
Adverse drug reactions
Adverse drug events
Sentinel events⁷
Prescribing errors may occur in:
Ordering/prescribing
Documentation
Transcribing
Dispensing
Administering
Monitoring
In a healthcare organization this translates into vulnerabilities for information systems, physicians, nurses, pharmacists and support staff. The result can be government intervention, e.g., state medical board and federal OIG⁸ investigations, malpractice claims, and insurer exclusions.
Common prescribing issues also include:
Deteriorated drugs from compromised storage
Drug utilization error from the administration, dispensing or monitoring of drugs
Human prescribing errors and omissions
In addition to storage and human error, prescribing errors can be compounded systemically. Prescribing errors traced to system failures include⁹:
Inaccurate order transcription
Drug knowledge dissemination
Failure to obtain the patient’s allergy history
Incomplete order checking
Medication tracking errors
Poor professional communication
Inaccurate or missing patient information
“Upwards of 75% of medication errors result from distraction.”
Obviously an incorrect dose on a prescription may have significant adverse consequences. Patient error or inappropriate patient action in taking a medication can be equally problematic, resulting in potential sentinel events¹⁰ and emergency room visits.¹¹
NCBI has identified distraction as a major cause of physician prescribing errors. Upwards of 75% of medication errors result from distraction, as a busy physician tries to fulfill multiple duties in a hospital, including patient rounds, ordering tests, examining patients, speaking with patient family members, and submitting insurance documentation. Many hospitals are attempting to reduce distraction-caused errors by requiring physicians to order medications at set times and instructing staff and consulting physicians not to disturb the prescribing doctor during this time.¹²
Prescribing error issues are not limited to the United States. The British Pharmaceutical Journal has identified the Top Ten Prescribing Errors in Practice (and how to avoid them).¹³ The Journal’s primary areas of concern are:
Omitted or delayed prescriptions
Incorrect use of anti-coagulants
Prescribing or use of opioids
Insulin and reactions
Prescribing of nonsteroidal anti-inflammatory drugs
Drugs that require blood test monitoring
Allergies to medications.
Drug interactions
Loading doses (multi-step drug calculations)
Oxygen when prescribed as a “drug” for hypoxaemic patients
This list is not exhaustive. It does, however, demonstrate the common, world-wide concern about accurate prescribing information, patient data, communications among professionals and patients, and data transmission. Having adequate check lists and cross-referencing of information from diagnosis through dispensing is therefore critical to avoiding errors.
The Problem With Prescribing Medications
Here in the U.S., NCBI cites incorrect dosages, allergy information errors, known contraindications, pharmacy errors due to illegible writing (fixable with digital technology) and the need to provide clear instructions to patients as crucial elements requiring review and correction through expanded use of technology and/or training. Risk analysis to reduce or prevent prescribing errors and exposure to liability has become especially important. Among risk factors for medication errors the NBCI includes:¹⁴
High volume
Poor handwriting
Inexperienced staff
Challenging patient populations
Lack of follow-up
Lack of appropriate monitoring
Lack of policy enforcement
Medically complex patients
Medications requiring calculations
Environmental factors
Poor communication
Shift work
Workplace culture
Verbal orders
Interpersonal factors such as external stress
Pharmacy Errors
Common pharmacy errors include delivery of an incorrect dosage, identifying contraindications, allergies, monitoring, drug interactions and knowledge deficits.¹⁵ Pharmacists, prescribing physicians, nurses and staff must function as a team to ensure these errors are avoided. Too often routine tasks such as prescribing become mundane and subject to error due to carelessness or fatigue. These errors can be corrected with proper review and training.
The Blame Game
NCBI also asks who is to blame for prescribing errors — the healthcare professional or the system?¹⁶ Medical Boards tend to blame the individual physician because that is who has a license with the Board for disciplinary action. The real “culprit” may extend to other employees due to:
Negligence
Forgetful
Hurrying
Poor motivation
Vengeance
Carelessness
In such situations, disciplinary actions may include:
Blame and shame
Loss of privileges
A threat of a medical malpractice lawsuit
Relief from certain duties
Transfer to another department
Today, there is less of a tendency to engage in the blame-game. Medication errors are viewed as a systemic problem with errors the result but not the cause of the problem. We need to understand how and why an error occurred, so that changes and corrections can be made. This is where a thorough, system-wide process review can result in significant improvement.
“Medication errors are viewed as a systemic problem with errors the result but not the cause of the problem. We need to understand how and why an error occurred, so that changes and corrections can be made. This is where a thorough, system-wide process review can result in significant improvement.”
The Kure for Prescribing Medications
Kure, with its Process Optimization Path™ or POP (Patent Pending) can provide an easy-to-use SaaS application that enables an individual staff person, physician, pharmacist or program managers to identify concerns, map out potential root causes, and develop workable solutions, without the need for outside audits, or bringing in expensive outside consultants or monopolizing staff time with specialized training in error-avoidance. Patents benefit, physicians can avoid disciplinary investigations, and hospitals can reduce liability exposure and government regulation.
How Kure Optimizes Prescribing Medications
As an example, suppose a physician examines a patient who presents with a severe muscle pain and determines that a new, relatively low-dose, Schedule III drug will be an effective treatment. We will call the drug Lavonka*. He tells the patient he is prescribing a Lavonka and that the patient should take it three times per day with food for two weeks. However, when his medical assistant calls in the prescription to the patient’s pharmacy in a neighboring community, the assistant reads the “3” in the 30 mg dose as an “8”. The pharmacy then receives a prescription for 80 mg of Lavonka, to be taken three times per day. This is an extraordinarily high dose for this drug and may cause severe side-effects leading to a sentinel patient event. Luckily, the pharmacist on duty is familiar with Lavonka and its usual dosage, and takes the step of calling the physician at the hospital to confirm the dose. A potential patient crisis was avoided. How could this have been prevented?
As the NCBI has stated with respect to prescribing issues:
“ . . The best method to enhance patient safety is to develop a multi-faceted strategy for education and prevention. Emphasis should be put on healthcare providers working as a team and communicating as well as encouraging patients to be more informed about their medications. With a culture of safety, dispensing medication errors can be reduced.¹⁷”
We agree with NCBI that developing a team approach to implement a multi-faceted strategy to reduce prescribing errors is critical to the mission of delivering safe, high-quality medical services to U.S. citizens. Join Kure to find solutions through optimization.
Supercharge your Lean Six Sigma projects and create Ethical Efficiency™ with Kure.
¹ CURES (Controlled Substance Utilization Review and Evaluation System) is a data platform maintained by the California Department of Justice that tracks all Schedule II-V controlled substances dispensed to patients. It includes prescribing data pertaining to physicians, pharmacists and patients, including patient names, date of birth, address, prescriber, DEA number, pharmacy with license information, the substance prescribed, date dispenses, prescription number, drug name and strength and refills remaining.
² Press Release, December 22, 2015.
³ As many as 64 California were accused of improper prescribing under the Death Certificate Program in 2019. Following extensive criticism of the program due to its broad scope, the Death Certificate Program has been revised and narrowed.
⁴ See “Medication Dispensing Errors and Prevention,” Rayhan A. Tariq, Rishik Vashisht, Ankur Sinha, NCBI Resources, July 25, 2021.
⁵ NCBI, Medication Dispensing Errors and Prevention, July 25, 2021.
⁶ Id.
⁷ The Joint Commission describes a sentinel event as “an unexpected occurrence involving death or serious physical or psychological injury, or the risk thereof”. Id.
⁸ Office of Inspector General, Department of Health and Human Services (HHS).
⁹ See footnote 1, supra.
¹⁰ A sentinel event is an unexpected occurrence involving death or serious physical or psychological injury.
¹¹ Id.
¹² Id.
¹³ See The Top Ten Prescribing Errors in Practice and How to Avoid Them.
¹⁴ See NCBI, supra, footnote 1.
¹⁵ Id.
¹⁶ See footnote 1, supra.
¹⁷ Id.
* Lavonka is a purely fictional drug name not related to any particular product or place.