Medical Materials Manufacturer Reduced Changeover Time By 66% With Kure
Project Summary
The problem: Long machine changeovers were creating unnecessary downtime and slowing production for a medical device manufacturer.
Using Kure’s structured project execution platform, the team reduced changeover time from 133 minutes to 45 minutes—an impressive 66% improvement.
Introduction
A leading medical materials manufacturer plays an important role in the healthcare supply chain by producing specialized materials used in medical devices and pharmaceutical applications. These materials support manufacturers who develop life-saving technologies, making quality, reliability, and consistency essential to daily operations.
Operating in such a demanding environment requires more than technical expertise. It also requires efficient production systems that allow teams to respond quickly to customer needs while maintaining strict quality standards.
When the organization noticed delays during equipment changeovers between product runs, leaders recognized an opportunity to improve operational efficiency. To address the challenge, the improvement team used Kure, a structured platform designed to help teams execute improvement projects with clarity and discipline.
By guiding the team through a step-by-step problem-solving process, Kure helped the organization analyze the issue, identify root causes, implement targeted improvements, and verify measurable results.
The project also reflected the principles of Ethical Efficiency™—improving processes in ways that respect people, reduce waste, and strengthen long-term performance.
The Problem
In medical materials manufacturing, production efficiency directly affects the companies that rely on those materials to build critical healthcare products. Delays at the materials stage can ripple throughout the supply chain.
At this organization, the time required to change equipment between production runs had become a growing challenge.
Each time the team switched from producing one material formulation to another, machines remained idle for an average of 133 minutes. During this time, operators prepared equipment, located tools, adjusted settings, and set up the next production batch.
While these steps were necessary, the process lacked consistency and organization. The extended downtime created several operational challenges:
Reduced equipment utilization
Slower production cycles
Increased pressure to meet customer timelines
In an industry where manufacturers depend on reliable materials supply, improving changeover efficiency became a priority for maintaining strong service and operational performance.
The Root Causes
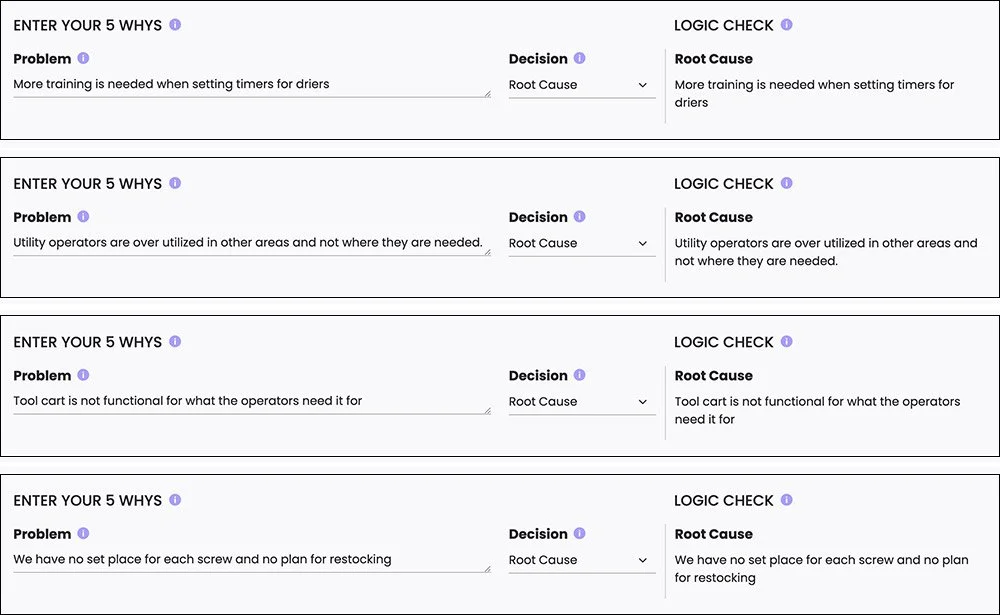
Using Kure’s structured problem-solving framework, the team analyzed the changeover process to understand what was contributing to the delays.
Their investigation revealed several key issues.
Utility operators were frequently assigned multiple responsibilities at the same time, which meant changeovers were sometimes understaffed and inconsistent.
Team members also lacked standardized training for certain tasks, such as setting timers on material dryers. Small uncertainties during these steps added time and slowed the process.
The team also discovered that a helpful internal guideline—avoiding breaks during active changeovers—was not consistently followed. When key team members stepped away, the process slowed significantly.
Finally, tools and components were not organized in a consistent way. Screws, parts, and equipment lacked designated storage locations, and the existing tool cart was not designed to support operator workflow during changeovers.
Using Kure’s 5 Whys root cause analysis, the team was able to move beyond surface-level symptoms and clearly identify the operational issues driving the delays.
The Solutions
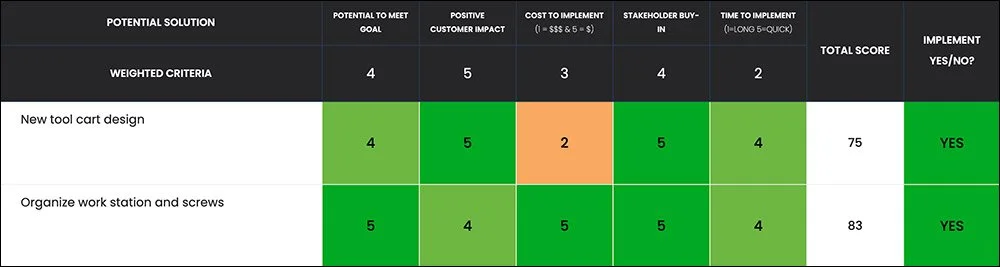
Once the root causes were confirmed, the team used Kure to organize and implement targeted improvements.
One of the most impactful changes was redesigning the tool cart based on operator feedback. The new design ensured the most commonly used tools were easy to locate and transport during changeovers.
Workstations were also reorganized to improve visibility and organization. Screws and small components were labeled and stored in clearly marked areas, and a consistent restocking plan ensured necessary supplies were always available.
Additional training helped ensure team members could confidently perform key tasks, including setting up and adjusting material dryers.
Leadership also reinforced the guideline of avoiding breaks during active changeovers so the team could maintain momentum until the process was complete.
Kure helped the team track these actions, document improvements, and ensure each solution directly addressed a confirmed root cause.
The Results
The improvements delivered a significant operational impact.
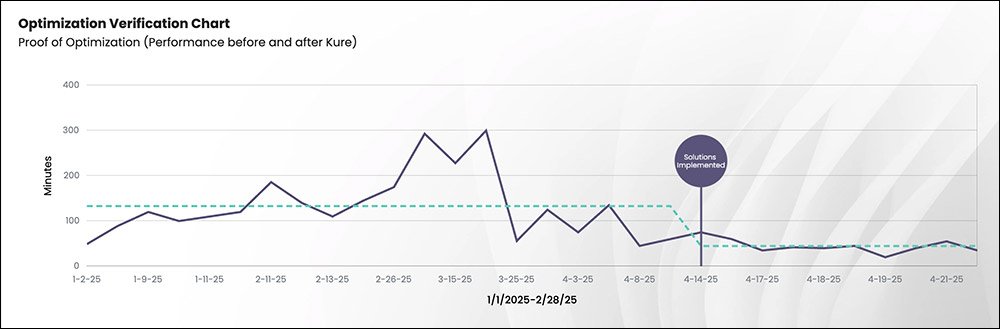
The team reduced changeover time from 133 minutes to 45 minutes, eliminating 88 minutes of downtime per changeover. This represents a 66% reduction in changeover time.
With faster changeovers, production lines return to operation sooner, helping the organization:
Increase equipment utilization
Accelerate production cycles
Improve delivery timelines for customers
Reduce unnecessary waste and idle time
Kure’s verification tools also allowed the team to clearly visualize the improvement through data, confirming the effectiveness of the new process.
Conclusion
This project demonstrates how structured execution and strong teamwork can deliver meaningful operational improvements.
By using Kure, the improvement team was able to move from identifying a problem to delivering verified results with clarity and discipline. The platform helped guide the team through root cause analysis, solution implementation, and performance verification—ensuring the project remained focused and effective.
Just as importantly, the project reflected the principles of Ethical Efficiency™. Rather than simply pushing employees to work faster, the team improved organization, training, and workflow so the process itself became more efficient and sustainable.
With a faster, more reliable changeover process in place, the medical materials manufacturer is better positioned to support its customers, improve production performance, and continue delivering high-quality materials that enable innovation across the healthcare industry.
*We value our clients’ confidentiality. While we’ve changed their names, the results are real.